Global MLR Process
In PromoMats Basics, the lifecycle that a document follows depends on the input for the Country and Product Type fields, and whether or not the Approved with Changes Country Setting has been enabled by an admin. To accommodate all relevant regulations, additional workflows and roles are available based on these selections.
A document can be Canceled if it has not reached a major version, i.e. v1.0 (meaning document has not yet been approved). A document’s previous major version can be Expired if Version is greater than 0 (meaning document has been previously approved). Document Version 1.0 becomes Superseded when version 2.0 becomes Approved for Distribution. Documents can be sent for Periodic Review for an Expiration Date extension. Documents do not change state during the evalution, but will end in a state that corresponds with the document’s Expiration Date.
United States
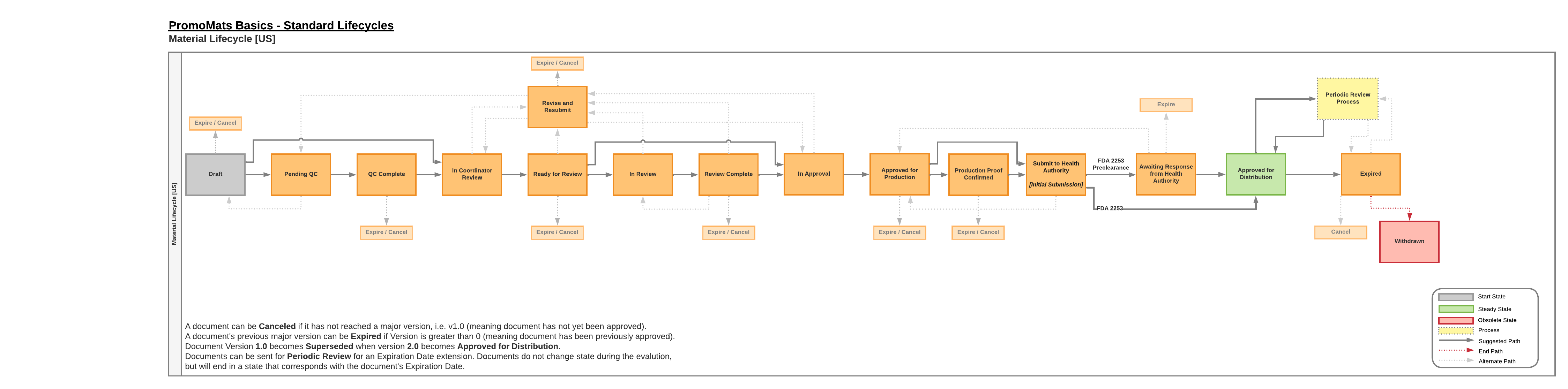
The document lifecycle for United States has the following defining features:
- Optional Review
- Users can bypass the Review workflow in favor of Approvals.
- Device Products
- Health authority submission fields and workflows are hidden, ad hoc reviewers have additional options in the Capacity field.
- Drug Products
- Utilize the Submit to FDA workflow after MLR approvals.
- Approved with Changes
- An admin can enable the Approve with Changes Country Setting, allowing the Approved with Changes verdict during the approval workflow.
Global, Italy, & Spain
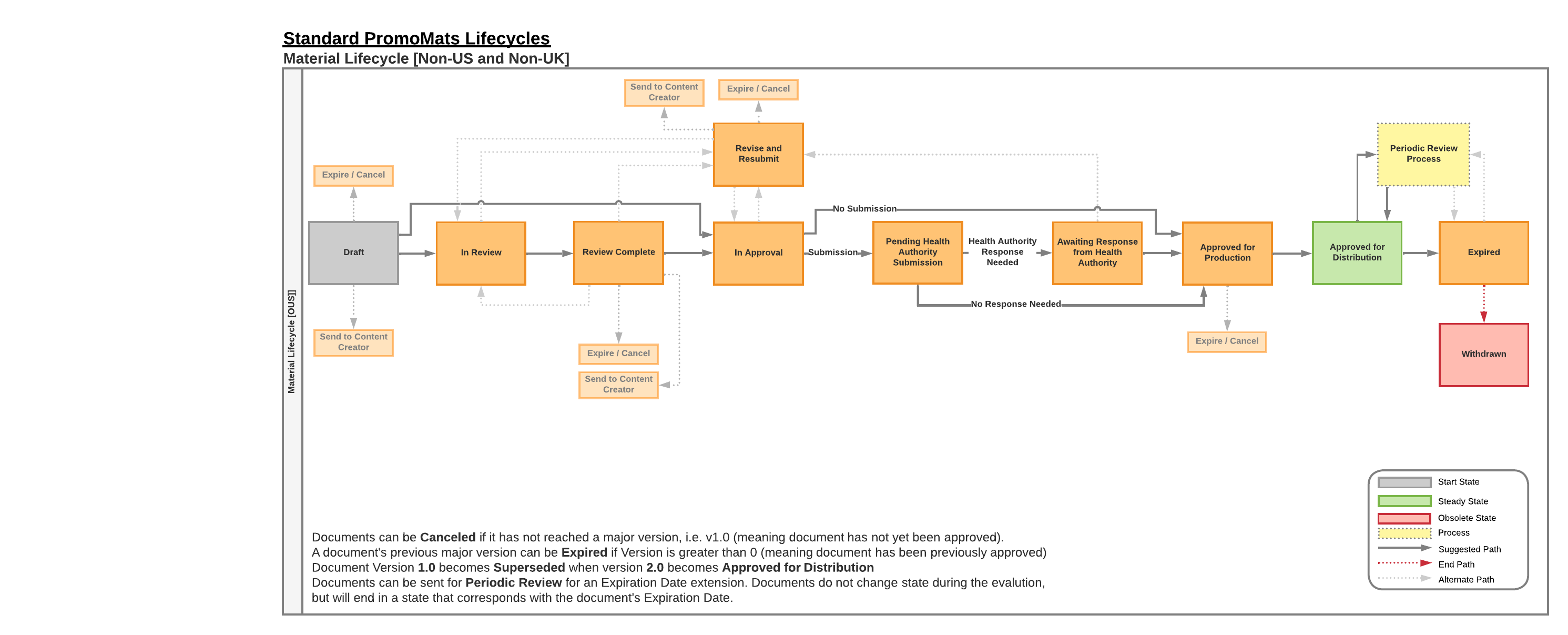
The document lifecycle for Global, Italy, & Spain has the following defining features:
- Optional Review
- Users can bypass the Review workflow in favor of Approvals.
- Device Products
- Health authority submission fields and workflows are hidden, ad hoc reviewers have additional options in the Capacity field.
- Drug Products
- Utilize the relevant health authority in the “Submit to…” workflow following MLR approvals.
- Approved with Changes
- An admin can enable the Approve with Changes Country Setting, allowing the Approved with Changes verdict during the approval workflow.
France
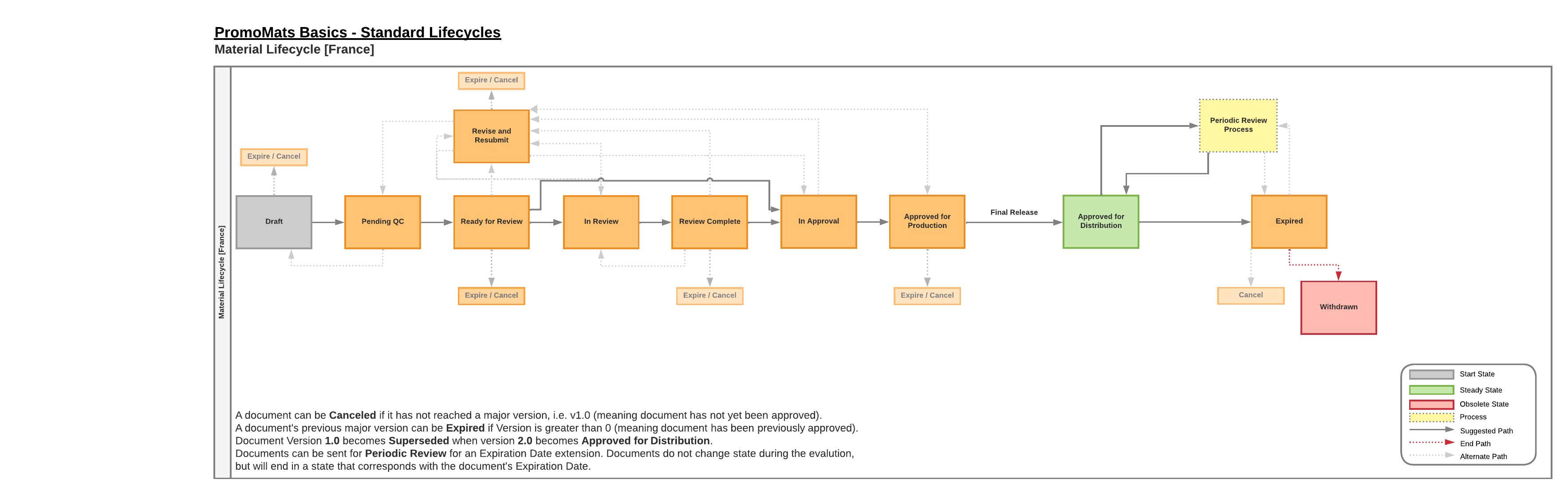
The document lifecycle for France has the following defining features:
- Optional Review
- Users can bypass the Review workflow in favor of Approvals.
- Drug Products
- Utilize the Final Release approval workflow featuring the Responsible Pharmacist role as the final approver.
- Device Products
- Utilize the same Final Release approval workflow, featuring the Person Responsible for Regulatory Compliance (i.e. a Regulatory user) as the final approver.
- Approved with Changes
- An admin can enable the Approve with Changes Country Setting, allowing the Approved with Changes verdict during the approval workflow.
United Kingdom
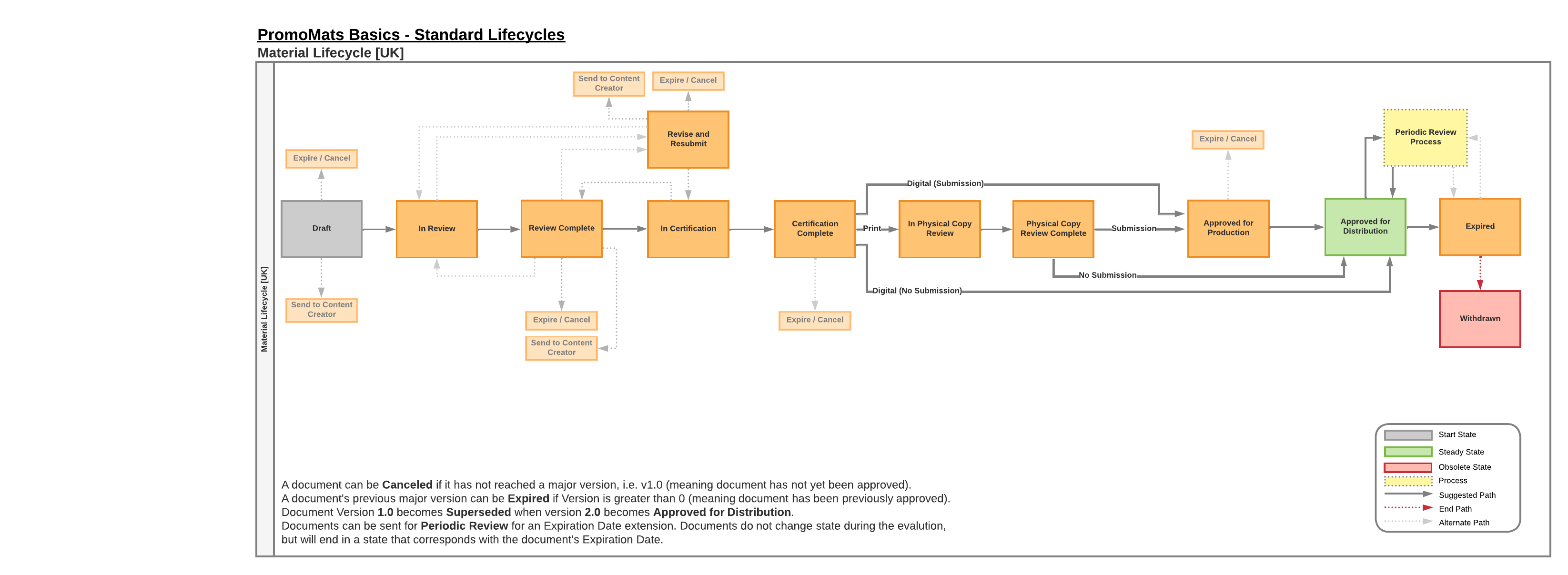
The document lifecycle for the United Kingdom has the following defining features:
- Media Field
- A required field on United Kingdom documents that states whether the final form of the material will be in print or digital format.
- Drug Products
- Utilize the Certification approval workflow for documents that require Medical Signatory and/or Appropriately Qualified Person (AQP) approval.
- If the Media field is set to Print, the Review Physical Copy workflow should take place after Certification.
- Utilize the Examination approval workflow for low-risk documents, where the approvers are traditional Medical, Legal, and Regulatory users.
- Device Products
- Follow a conventional MLR process with Medical, Legal, and Regulatory users providing review and approval.
- Approved with Changes
- An admin can enable the Approve with Changes Country Setting, allowing the Approved with Changes verdict during the approval workflow for Device Products (does not apply to Certification and Examination).

